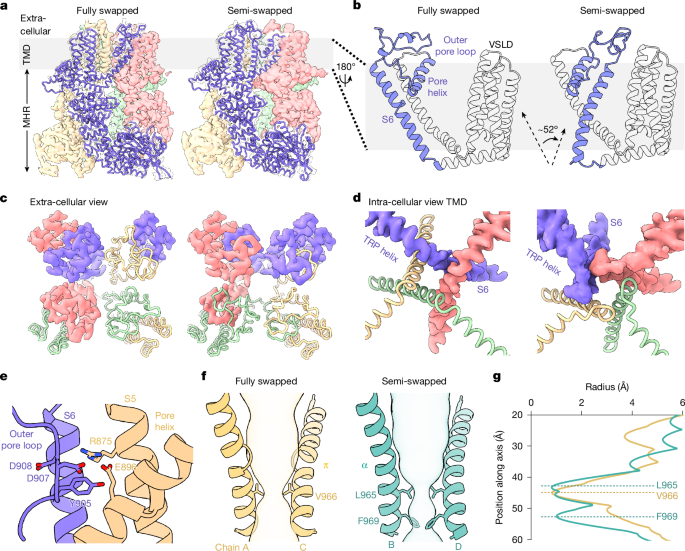
Structural energetics of cold sensitivity
Key Points:
- Researchers engineered N-terminally fused mEGFP TRPM8 constructs via Gibson assembly and expressed them in Expi293F cells using baculovirus transduction, followed by sodium butyrate supplementation to boost protein expression.
- TRPM8 purification involved detergent solubilization with LMNG/GDN, affinity capture using anti-GFP nanobody resin, protease cleavage, and size-exclusion chromatography, with protocols adapted for different pH conditions and sample types (detergent vs. vesicle).
- Cryo-EM grid preparation and data collection were performed using various holey carbon grids and state-of-the-art microscopes (Titan Krios, Glacios), with datasets processed through cryoSPARC and RELION pipelines to generate high-resolution TRPM8 structures.
- Hydrogen–deuterium exchange mass spectrometry (HDX–MS) was utilized to probe TRPM8 dynamics and thermodynamics, revealing EX2 exchange kinetics that allowed quantification of folding free energy changes upon menthol binding and temperature variations.
- Functional calcium imaging assays using Fura-2-AM in transfected HEK293T cells assessed TRPM8 activation by cold and menthol, while sequence alignments of avian and mammalian TRPM8 orthologues provided evolutionary context for structural findings.







.jpg?width=1200&height=630&fit=crop&enable=upscale&auto=webp)




