Check Your Medicine Cabinet for These Eye Drop Bottles After Millions Recalled
Key Points:
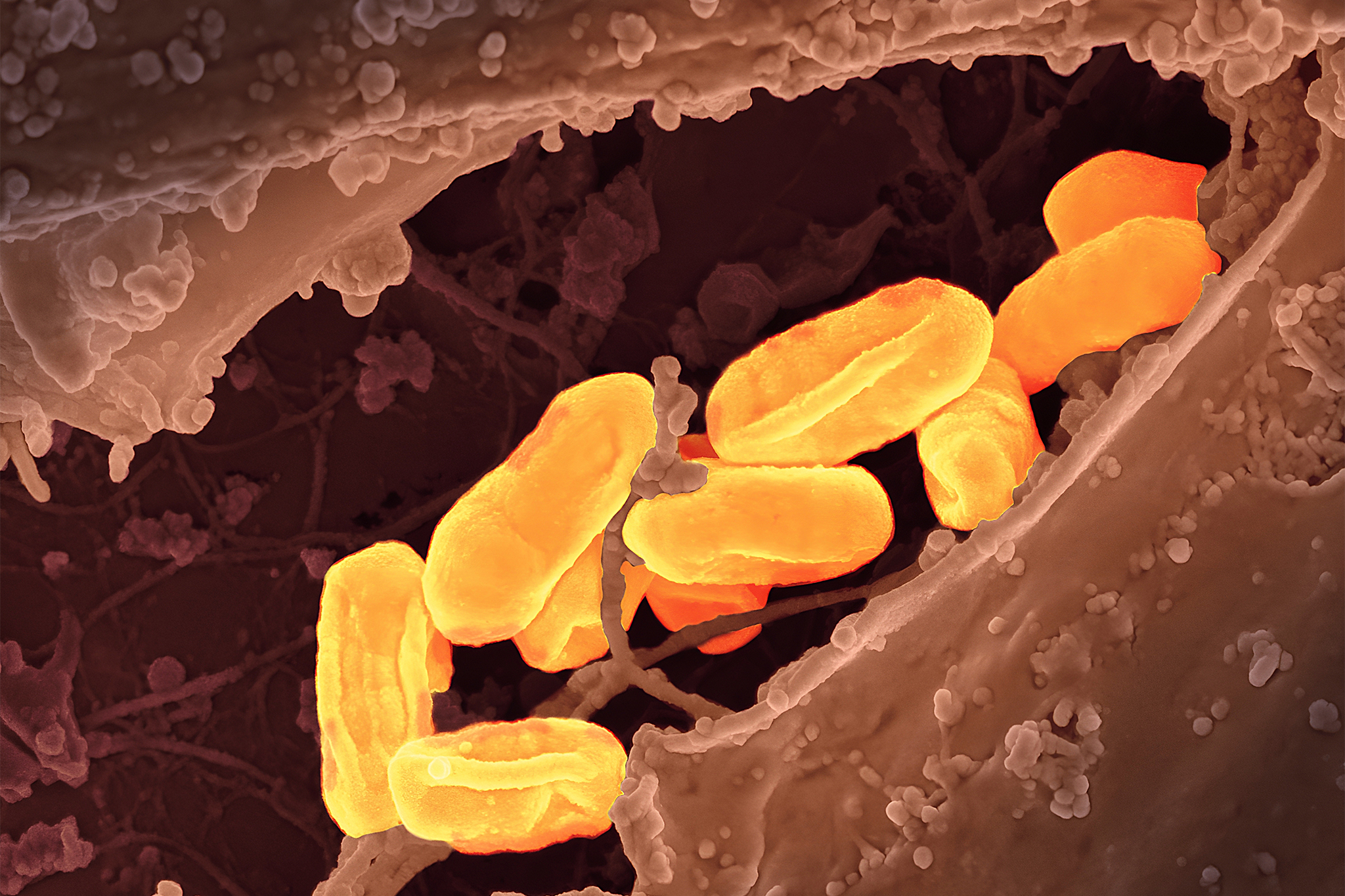
- Over 3 million bottles of eye drops manufactured by K.C. Pharmaceuticals in Pomona, California, have been recalled due to sterility concerns, affecting multiple brands sold at major U.S. drug stores and supermarkets.
- The recall covers eight main categories of eye drops, including Sterile Eye Drops A.C., Advanced Relief, Dry Eye Relief, Ultra Lubricating, Original Formula, Redness Lubricant, Soothing Tears, and Artificial Tears Sterile Lubricant.
- Popular retailers impacted by the recall include Walgreens, CVS, Kroger, Rite Aid, Meijer, H-E-B, and others, with many products distributed under various store and brand names.
- The FDA has not issued a press release about the recall, and K.C. Pharmaceuticals has not responded to inquiries regarding guidance for consumers on handling the recalled products.
- Consumers can find the complete list of recalled products and their UPC numbers on the FDA’s website to identify affected eye drops.










.png)