Johns Hopkins Team Develops Therapeutic, Nasally-Delivered DNA Vaccine for Tuberculosis
Key Points:
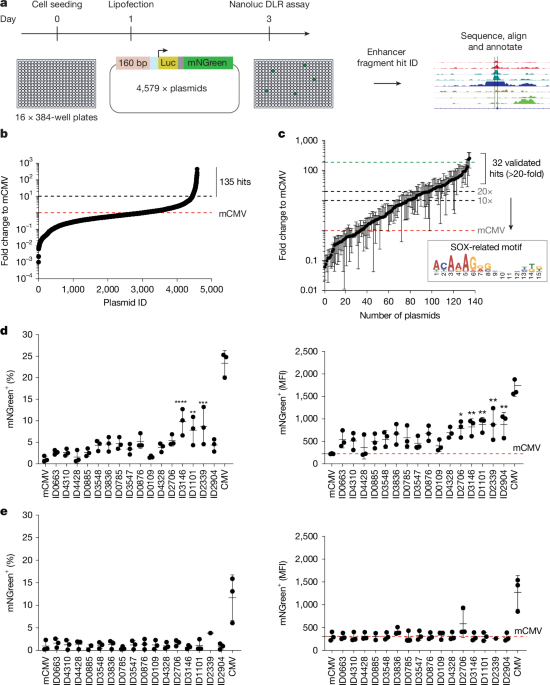
- Johns Hopkins Medicine researchers have developed a nose-delivered DNA vaccine targeting tuberculosis (TB), aiming to enhance immune response against drug-tolerant TB bacteria that survive antibiotic treatment.
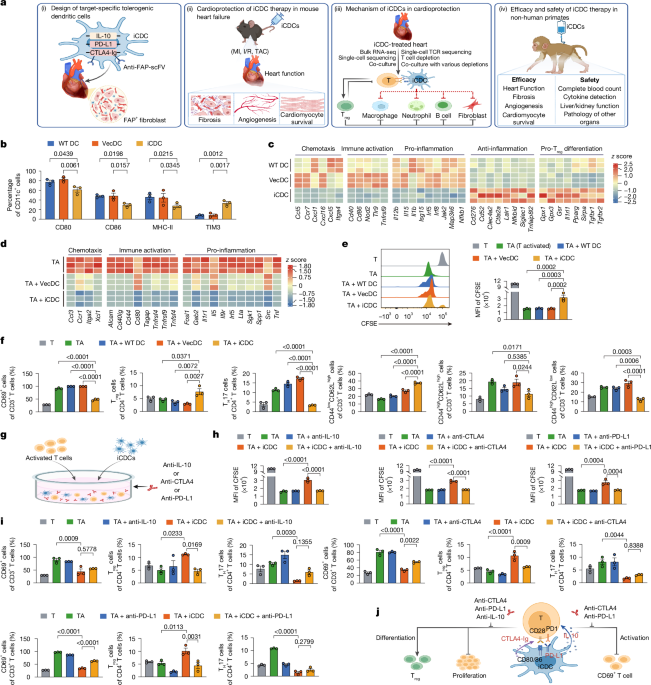
- The vaccine fuses two TB genes, relMtb and Mip3α, to stimulate dendritic cells and T-cells in the respiratory tract, improving bacterial clearance, reducing lung inflammation, and preventing relapse in mouse models.
- In studies involving rhesus macaques, the vaccine generated durable immune responses in blood and airways for at least six months, suggesting potential effectiveness in humans due to the primates' immune similarity.
- This approach aligns with WHO calls for therapeutic vaccines to complement drug therapies, especially to shorten treatment duration and combat drug-resistant TB strains.
- DNA vaccines offer practical benefits such as stability and efficient manufacturing, supporting their potential as a viable immunotherapy strategy alongside antibiotics for TB control.